Hiding in plain sight: Hines group uncovers new class of medicinal compounds that target RNA

A team of undergraduate and graduate chemistry students in Jennifer Hines' lab recently uncovered a new class of compounds that can target RNA and disrupt its function. This discovery identified a chemical scaffold that could ultimately be used in the development of RNA-targeted medicines to treat bacterial and viral infections, as well as cancer and metabolic diseases.
RNA is chemically like DNA but also controls the extent to which the DNA’s instructions are carried out within a living cell. It is this essential regulatory role in the function of the cell that makes RNA such an attractive target.

“Trying to target RNA is at the forefront of medicinal chemistry research with enormous potential for treating diseases,” said Hines, professor of chemistry and biochemistry in the College of Arts and Sciences. “However, there are relatively few compounds known to directly modulate RNA activity which makes it challenging to design new RNA-targeted therapeutics.”
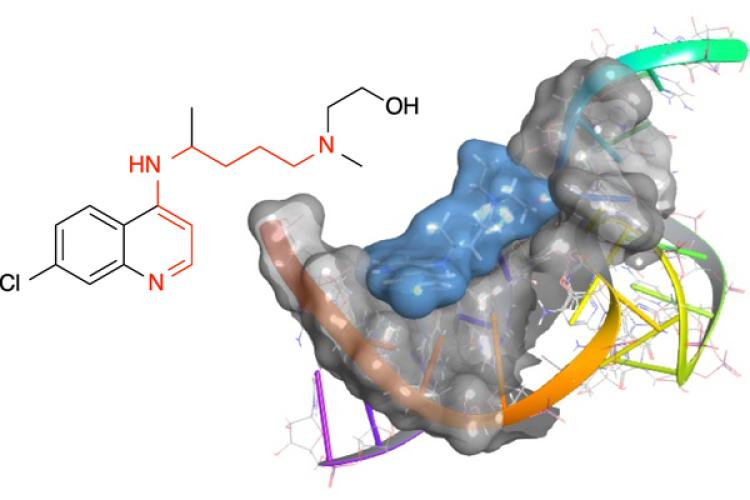
The Hines group determined that 4-aminoquinolines inhibit the function of the bacterial T-box riboswitch RNA and bind the stem-loop II motif RNA (an RNA structure found in the virus causing the COVID-19 pandemic).
“The compounds bind these RNA structures in very specific sites, making them good starting scaffolds for designing specific therapeutics. What was so surprising about this discovery is that the likelihood for RNA binding was hiding in plain sight within the 4-aminoquinoline structure, but no one had identified it before,” Hines said. “Our research determined that 4-aminoquinolines have distinct activities and chemical features very similar to polyamines which are natural compounds in the cell that modulate RNA function.”

Co-first authors on the paper were graduate student Md. Ismail Hossain and undergraduate student Mason Myers, who graduated from OHIO in 2021 with a B.S. in Chemistry from the Honors Tutorial College (HTC). Myers is now a doctoral student in chemistry and biochemistry at the University of Michigan. Additional authors were graduate students Danushika Herath and Ali Aldhumani, along with undergraduate student Hannah Boesger, who graduated in 2022 with a B.S. in Neuroscience from HTC as well. Boesger is now a doctoral student in medicinal chemistry at the College of Pharmacy at the University of Michigan.

"As part of a comprehensive RNA-targeted drug discovery project, we have been focused on investigating ligand-RNA binding interactions involving larger, more dynamic RNA structural motifs for more than 20 years. This experience is what enabled my group to so quickly respond, when the pandemic began, to investigate targeting the viral stem-loop II motif RNA first virtually via computational studies and then in the lab," Hines said.
The Hines group uses a combination of spectroscopic (fluorescence, UV-Vis, NMR); biochemical/biophysical (gel electrophoresis, isothermal titration calorimetry); and, computational (docking, molecular dynamics simulations, quantitative structure activity calculations, bioinformatics) techniques in their RNA-targeted drug discovery studies.
"It was in this earlier study where we first noticed the 4-aminoquinolines, but not enough was known about the function of the stem-loop II motif RNA to discern what the compounds might be doing," Hines added. "Consequently, we shifted to exploring the functional effect of these compounds on the T-box riboswitch RNA, which regulates gene expression in bacteria. In these riboswitch studies, we found that the compound’s inhibitory effect was dose-dependent in a manner very similar to the dose-dependency of polyamines, a class of compounds that normally bind RNA in the cell. It was in puzzling out why this might be the case when I noticed the structural similarity between the two classes of compounds."
The research, funded by the National Institutes of Health, was published online Dec. 29, 2022, in Biochemical and Biophysical Research Communications (4-Aminoquinolines modulate RNA structure and function: Pharmacophore implications of a conformationally restricted polyamine) and appeared in print in the Feb. 12, 2023, issue.

